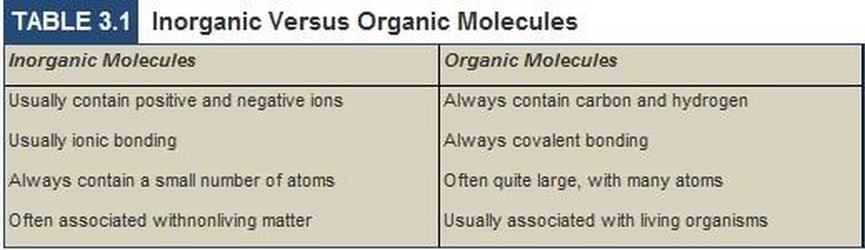
Organic vs. Inorganic Molecules
In the simplest of terms, a molecule is considered 'organic' if it contains Carbon and Hydrogen. If it does not have at least these two elements, it is considered 'inorganic'.
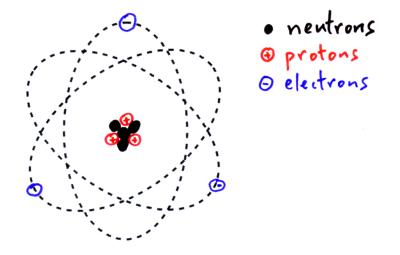
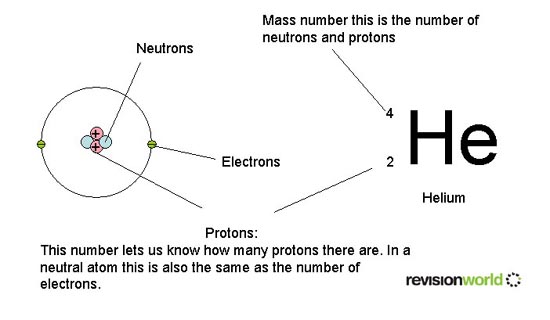
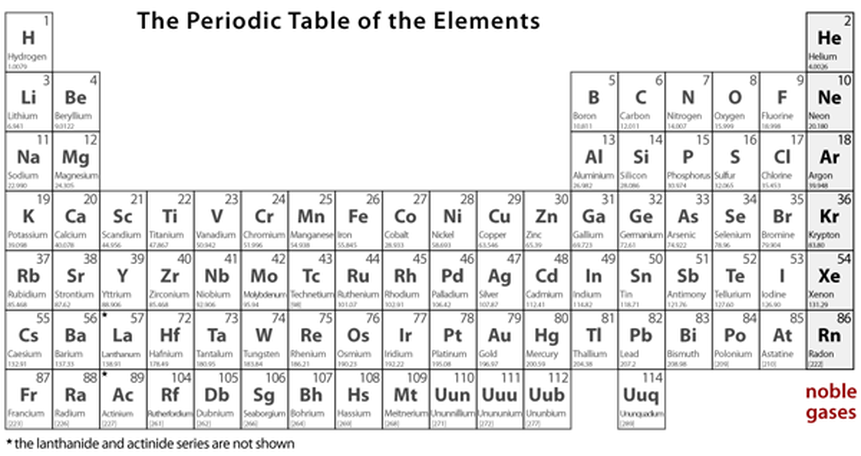
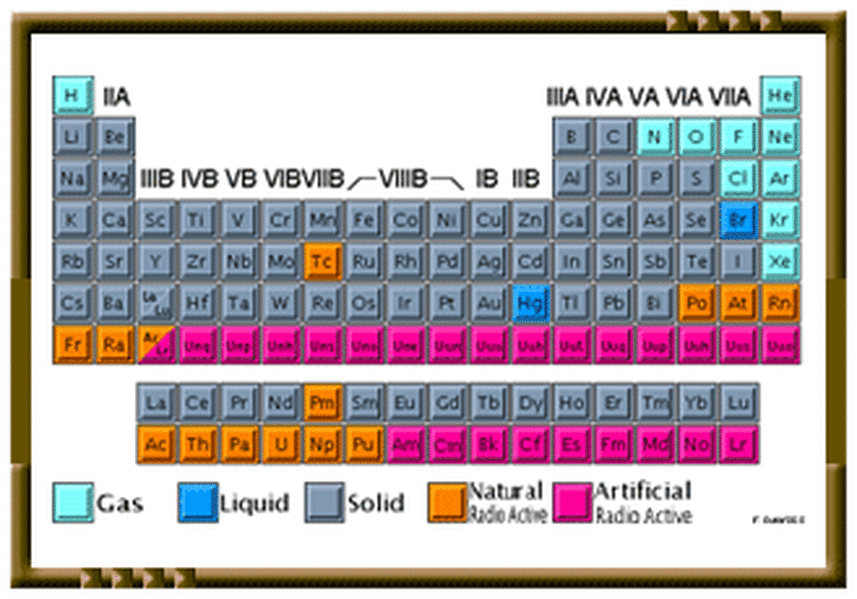
The Atomic Structure & The Periodic Table
Structure of an atom: protons, neutrons, and electrons.
|
Element in the Periodic Table of Elements:
|
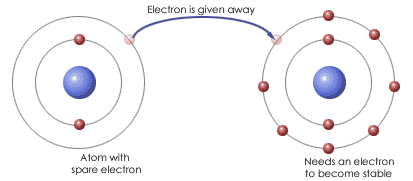
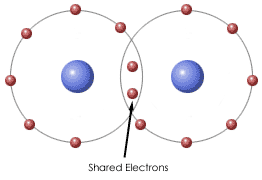
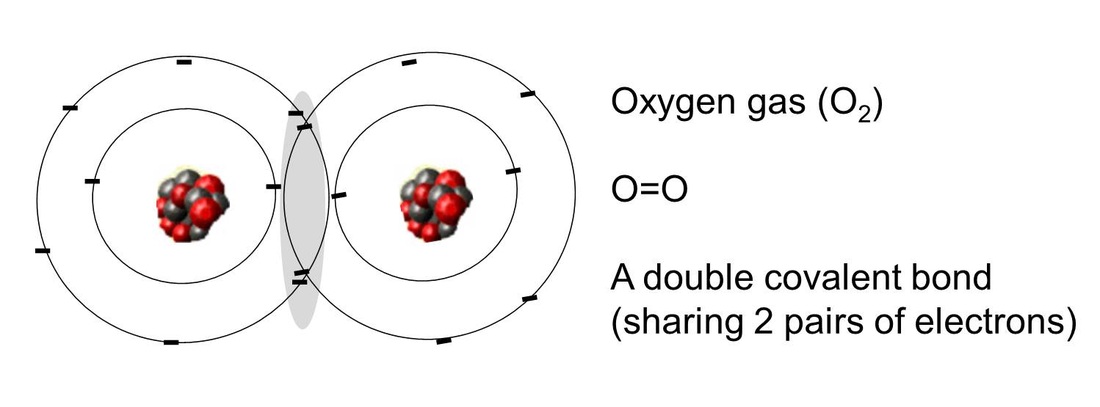
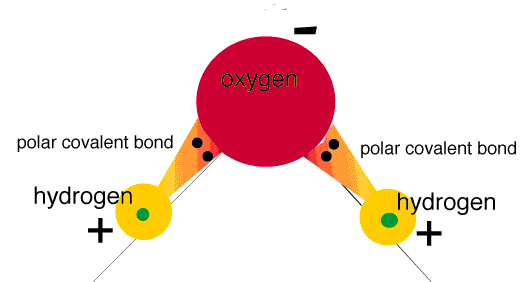
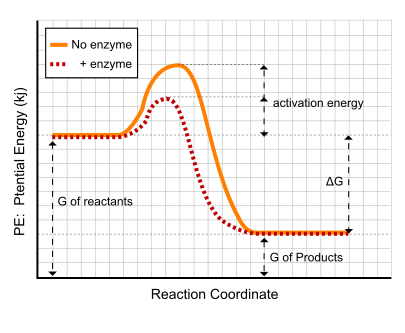
Bonding- a bond is formed when two atomic nuclei attract the same electron(s).
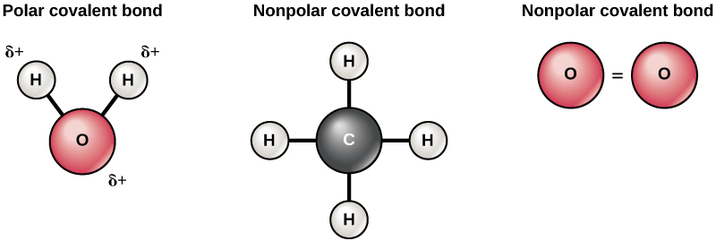
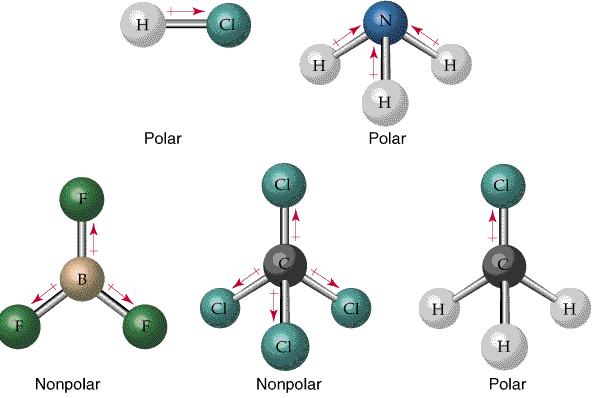
Polar covalent bond: electrons are shared unequally |
Intermolecular Attractions: attractions BETWEEN molecules
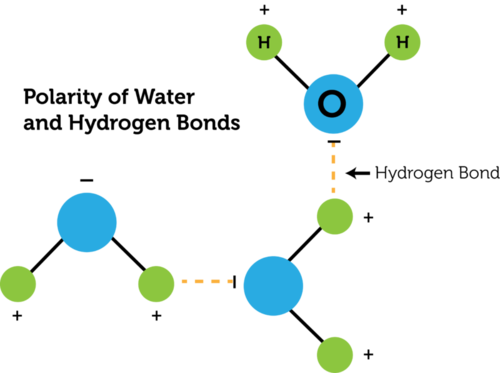
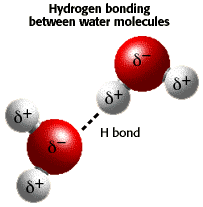
Hydrogen Bond: bond formed between hydrogen with Florine, Oxygen, or Nitrogen (between molecules.. an intermolecular force)
Characteristics of Water:
Water is very POLAR. It has STRONG intermolecular attractions due to its polar attractions and hydrogen bonding.
- Water has high specific heat: amount of heat that must be absorbed in order to raise 1 gram of a substance 1 degree Celsius. (Ex: large bodies of water [the ocean] absorb heat and resist changes in temperature)...therefore, they are stable environments for organisms.
- Water has a high heat of vaporization: a relatively great amount of heat is needed to evaporate water; evaporation of sweat significantly cools the body surface
- Water has high adhesion properties: forces of adhesion contribute to capillary action, helping water flow up from the roots of a plant to the leaves
- Water is the universal solvent: since water is highly polar, it is dissolves all polar and ionic substances
- Water exhibits strong cohesion tension: molecules of water tend to stick to each other. So, water can move up a plant without expending any energy; insects can 'walk on water'
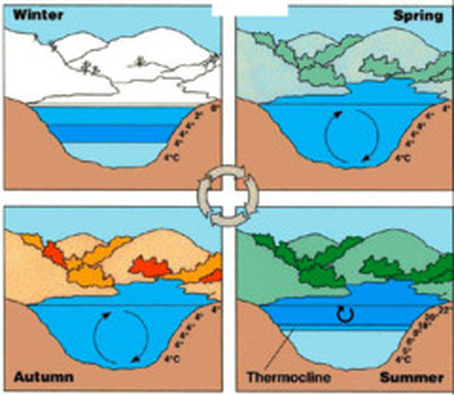
- Ice floats because it is less dense than water: floating ice insulates the water below it, allowing life to exist beneath a frozen surface. When ice melts, it sinks to the bottom of a lake, resulting in circulating water. Oxygen from the surface returns to the bottom of the lake, and nutrients from the 'bottom-dwelling' bacteria are carried to the surface. This "SPRING OVERTURN" is necessary for the health of a lake.
Isotopes: atoms of one element that vary only in the number of neutrons in the nucleus.
|
Isotopes: versions of an atom with varying number of neutrons.
|
|
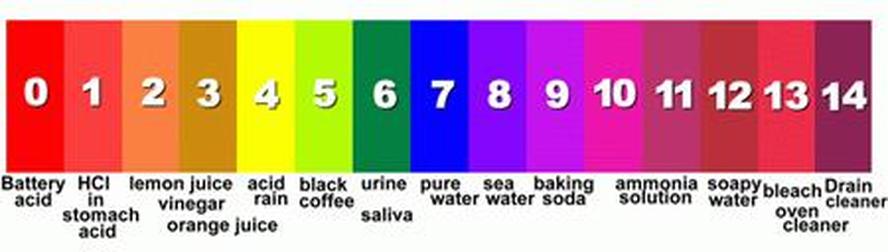
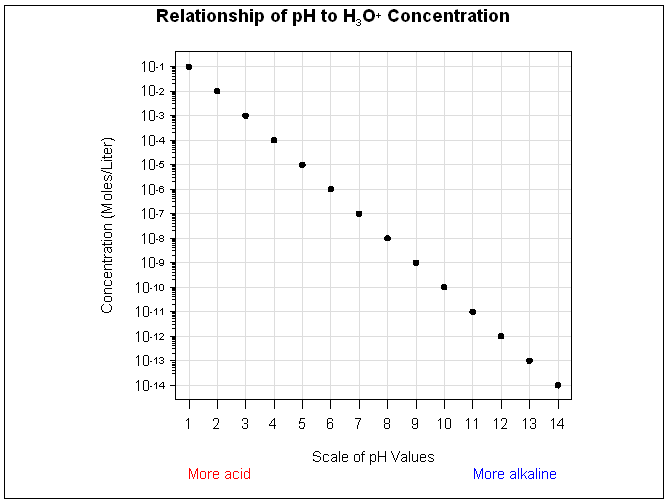
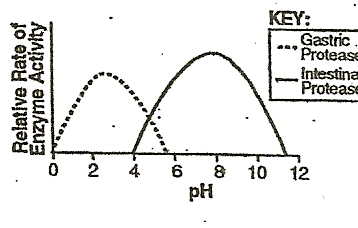
pH: the measure of acidity and alkalinity of a solution.
pH is a measure of the amount of Hydrogen ion concentration in moles per liter in a negative logarithm. A solution with a pH of 1 is 10x more acidic than a solution with a pH of 2, and 100x more acidic than a solution with a pH of 3.
|
Fun Facts
|
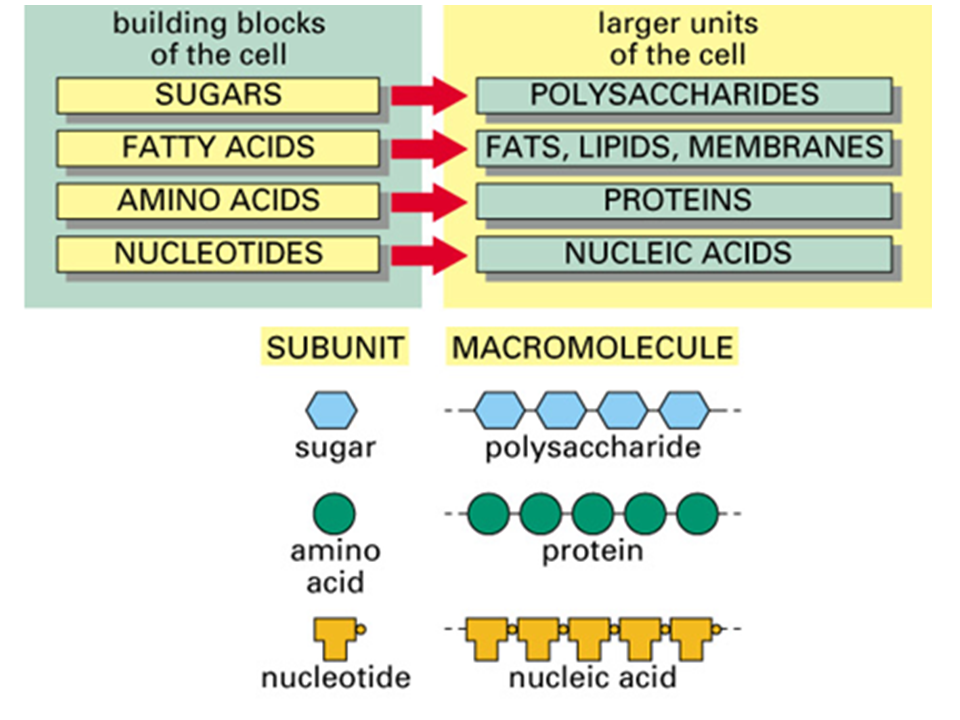
Organic Compounds
4 Macromolecules: Polysaccharides, Lipids, Proteins, Nucleic Acids
Polymers are made up of repeating monomers (building blocks or subunits)
1. Carbohydrates
- 3 elements: Carbon, Hydrogen, Oxygen
- Quick supply of energy
- 1 gram of carb will release 4 calories of heat when burned
- found in rice, pasta, bread, cookies
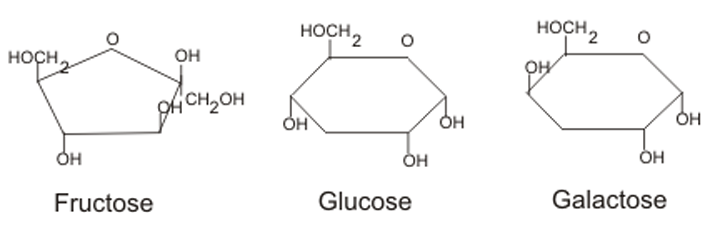
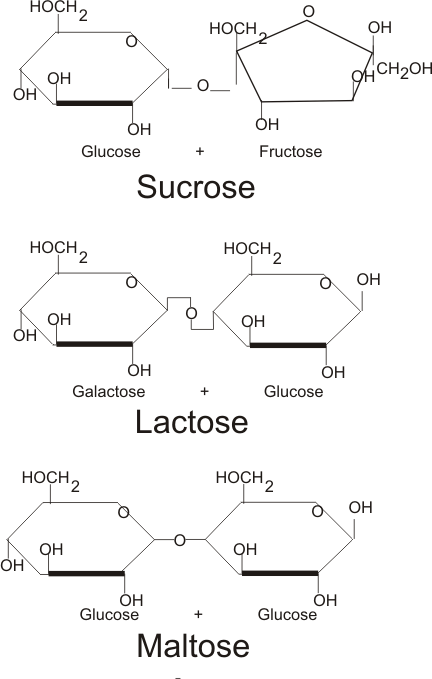
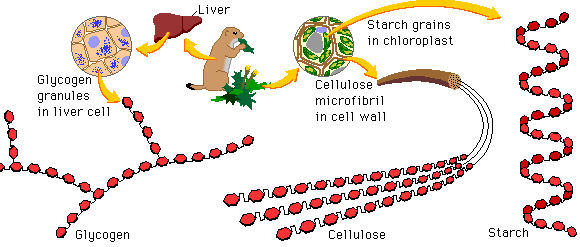
- 3 classes: monosaccharides, disaccharides, polysaccharides
Monosaccharides: C6H12O6
|
|
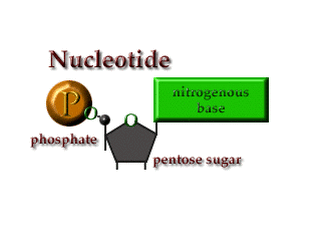
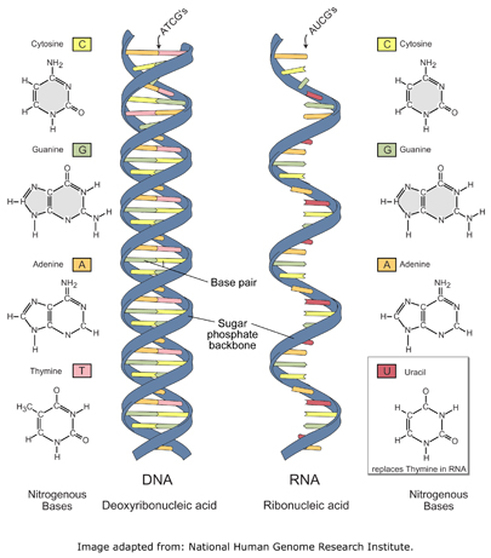
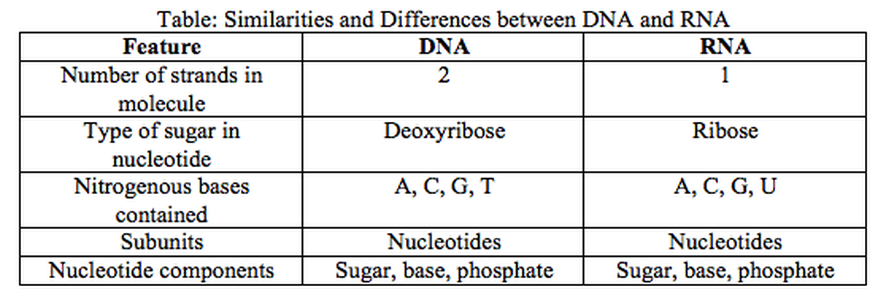
2. Nucleic Acids
3. Lipids
|
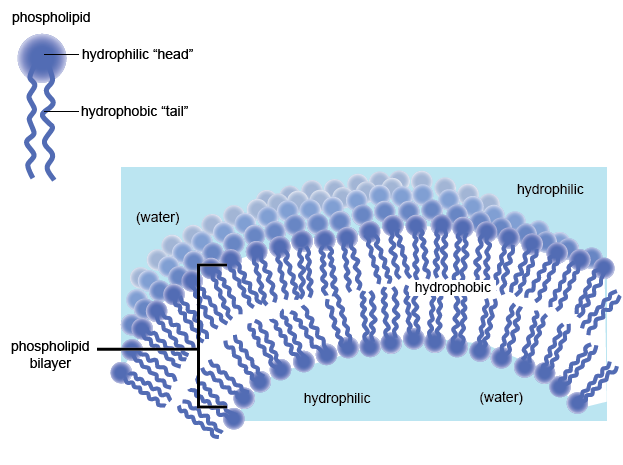
Hydrophobic ("water hating") vs Hydrophillic ("water loving")
Nonpolar substances will no dissolve in water; nonpolar substances are hydrophobic. Polar substances will dissolve in water; polar substances are hydrophillic NOTE: Like dissolves Like Lipids are nonpolar & hydrophobic. The plasma membrane only allows nonpolar substances to readily dissolve through the membrane; large polar molecules cannot diffuse across a plasma membrane. |
|
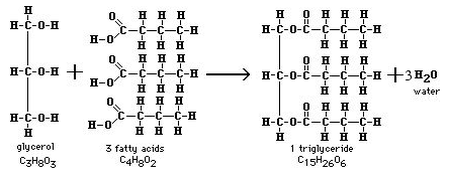
Lipids: fats, oils, waxes
|
|
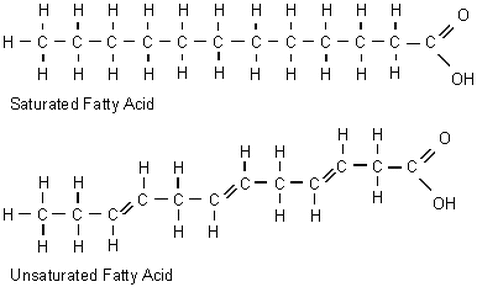
Saturated vs Unsaturated Fatty Acids:
|
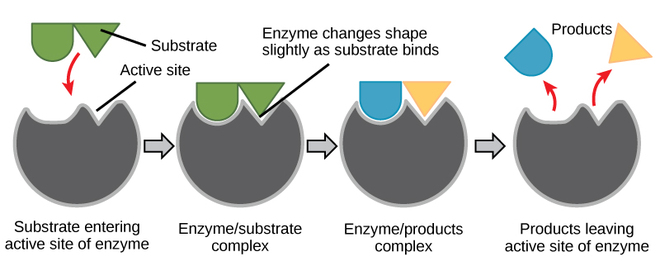
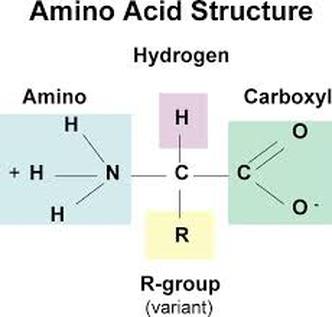
4. Proteins
|
polymer: Protein = polypeptide held together by peptide bonds
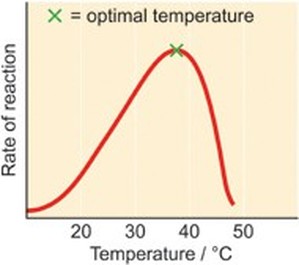
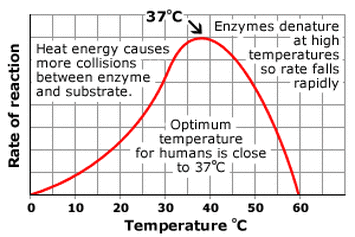
monomer: amino acids - a dipeptide: two amino acids held together by a peptide bond - proteins are responsible for growth and repair - dietary sources: fish, poultry, meat, beans, peanuts - 1 gram of proteins releases 4 calories of heat - Elements: S, P, C, O, H, N - only 20 amino acids exist, all differing in the R Group - enzymes are types of proteins |
|
|
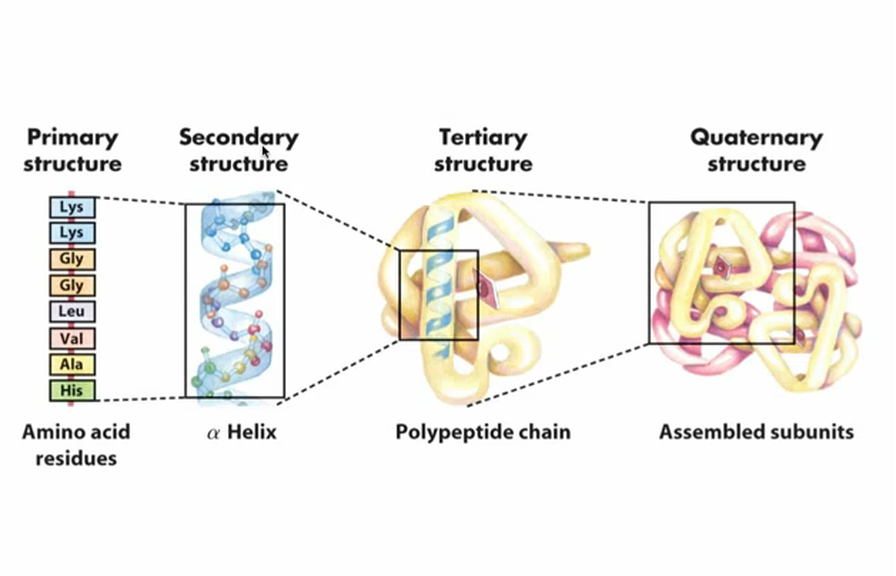
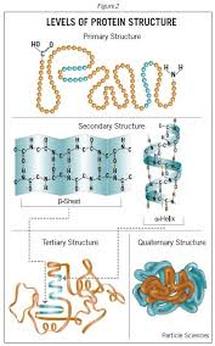
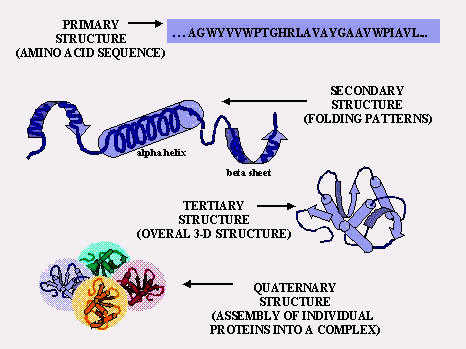
Primary: sequence of amino acids
|
Interesting fact: Prions- proteins that cause disease
Prions are infectious proteins, causing brain disease such as mad cow disease. Prions are misfolded version of a protein normally found in the brains of mammals. If a prion gets into a healthy brain, it causes normal proteins to misfold in the same way.
Notes: (* Notes and Powerpoints adapted from Shannan Muskopf's fantastic teaching resource: Biology Corner.)