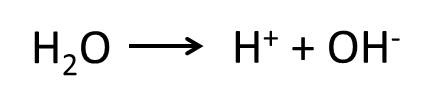|
|
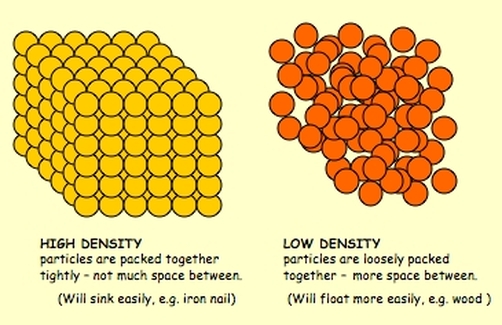
Density: the mass of matter per unit volume (grams/cubic centimeters)
|
Varying substances and their respective densities. The liquid is labeled on the left and the solid objects are labeled on the right:
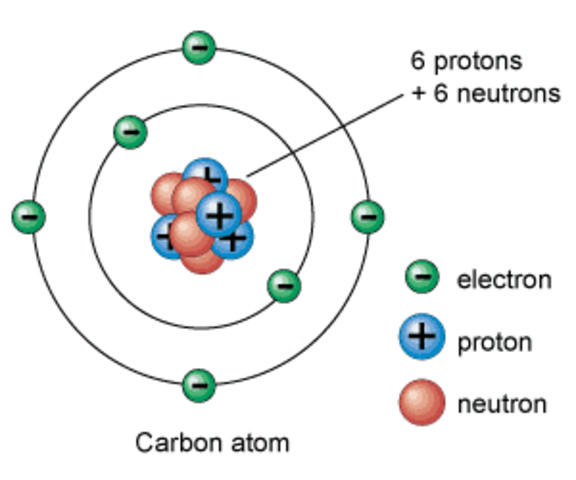
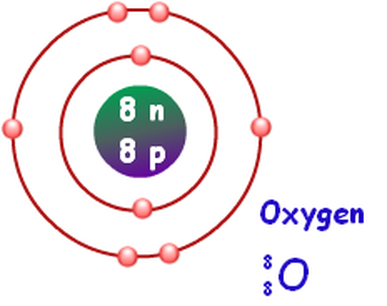
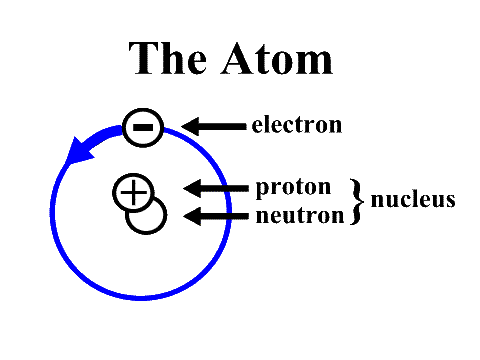
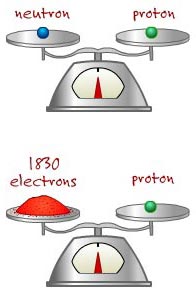
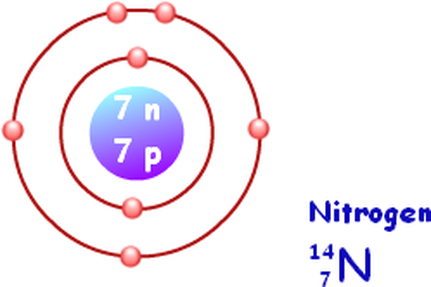
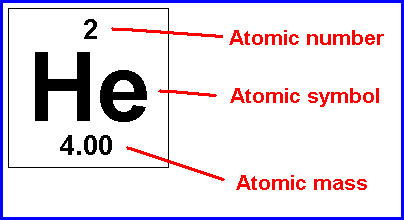
The ATOM:
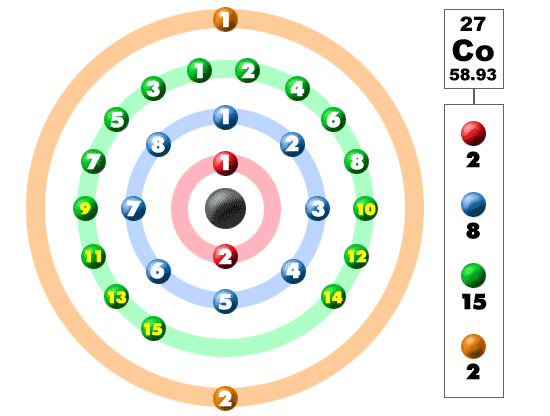
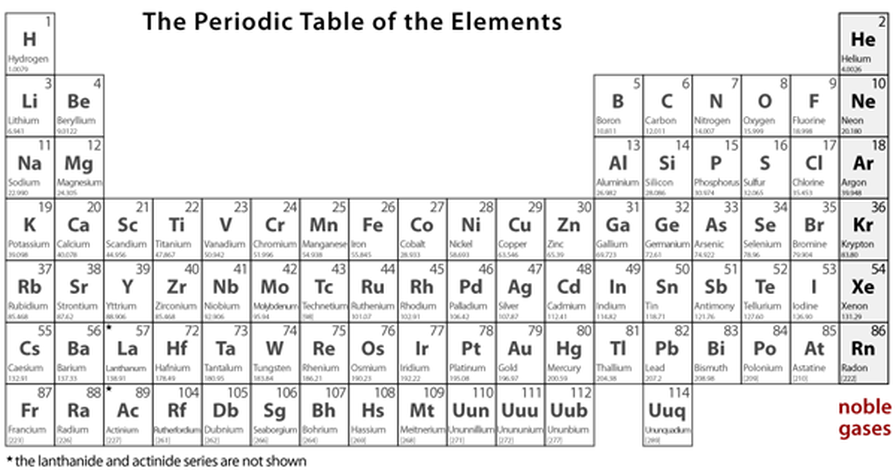
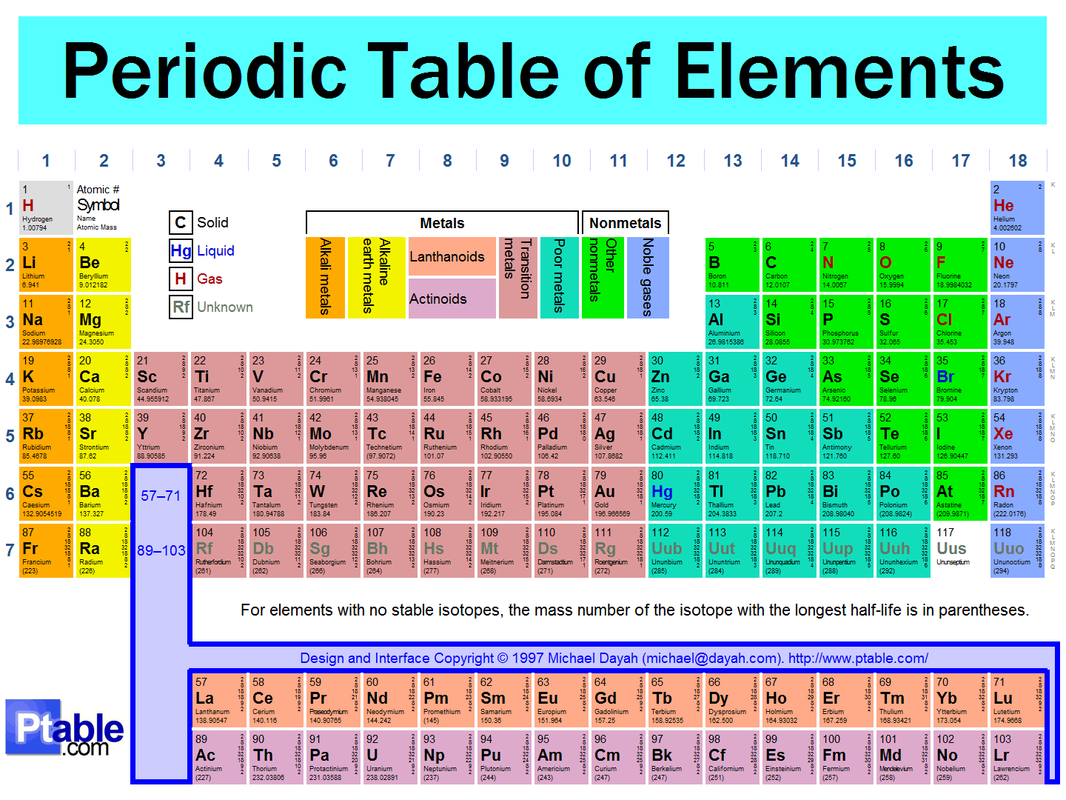
Periodic Table of Elements:
Bonding
|
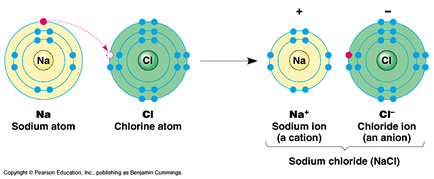
Ionic Bonding: a bond formed between two atoms that results in a transfer of one or more electrons. Each atom will have a charge, and therefore become an ion.
|
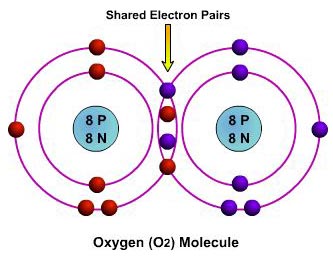
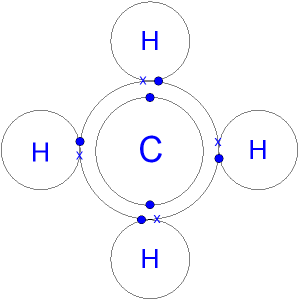
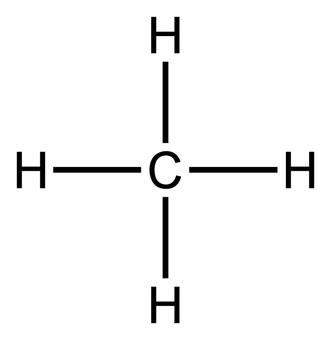
Covalent Bonding: a bond formed between two atoms that results in the sharing of two or more electrons.
|
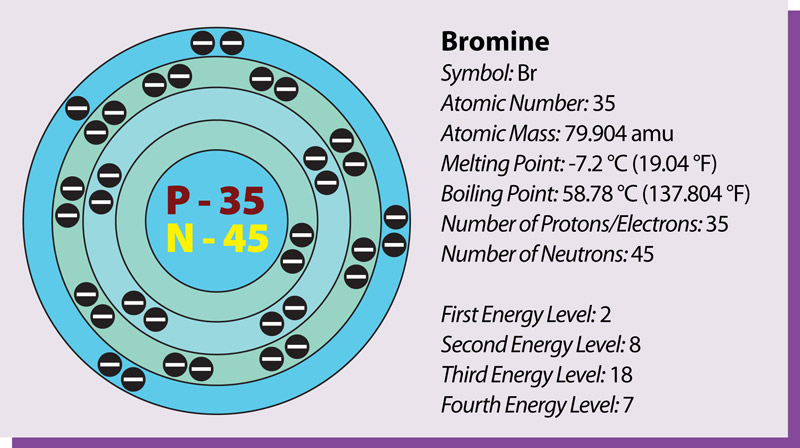
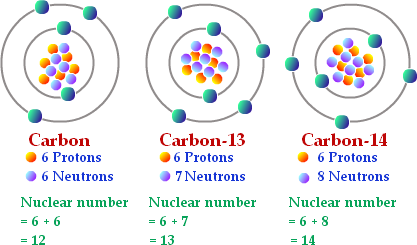
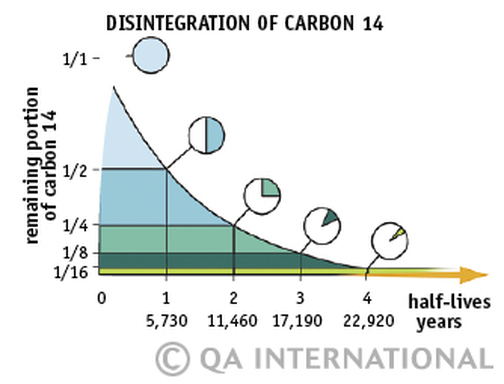
Isotopes
Isotopes are two or more forms of the same element with the same number of protons but different number of neutrons. Many isotopes are considered radioactive, meaning they are 'unstable' and will decay.
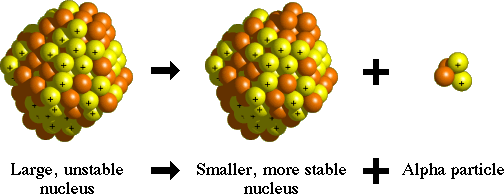
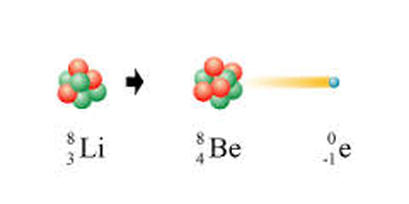
RADIOACTIVE DECAY::: release of particles in order to move from a high energy state back to a lower more stable energy state
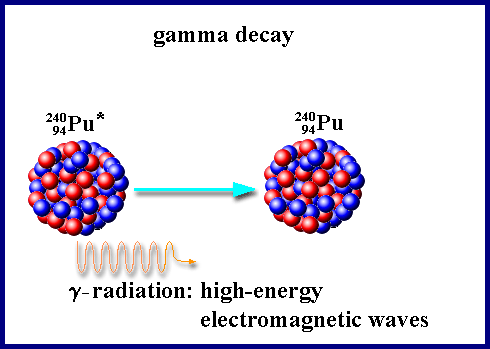
Gamma Decay: does not change the mass #, does not change the atomic #
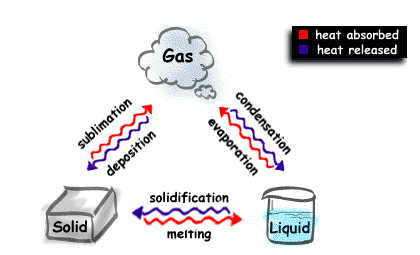
SOLIDS, LIQUIDS, and GASES...
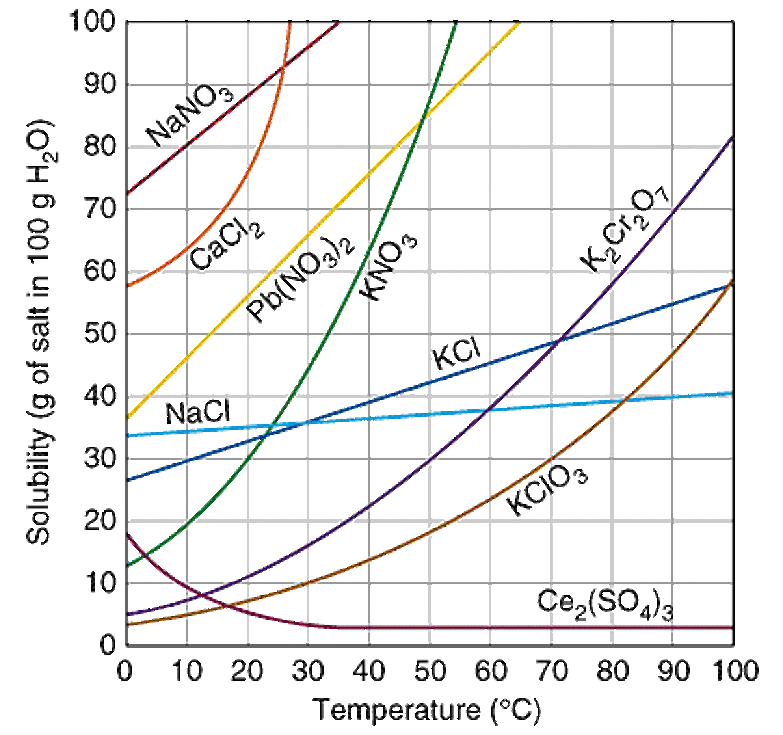
Solubility
Solubility is the property of a solid, liquid, or gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent to form a homogeneous solution of the solute in the solvent
Solute: the substance that is dissolved by the solvent
Solvent: the liquid that dissolves the solute
Solution: solvent + solute
Solute: the substance that is dissolved by the solvent
Solvent: the liquid that dissolves the solute
Solution: solvent + solute
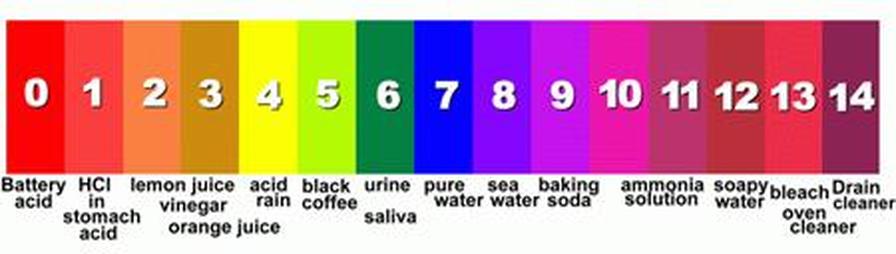
pH
Indicators are used to determine the pH of a substance
Red Litmus paper will turn Red to Blue in the presence of a base.
Blue Litmus Paper will turn Blue to Red in the presence of an acid.
Red Litmus paper will turn Red to Blue in the presence of a base.
Blue Litmus Paper will turn Blue to Red in the presence of an acid.